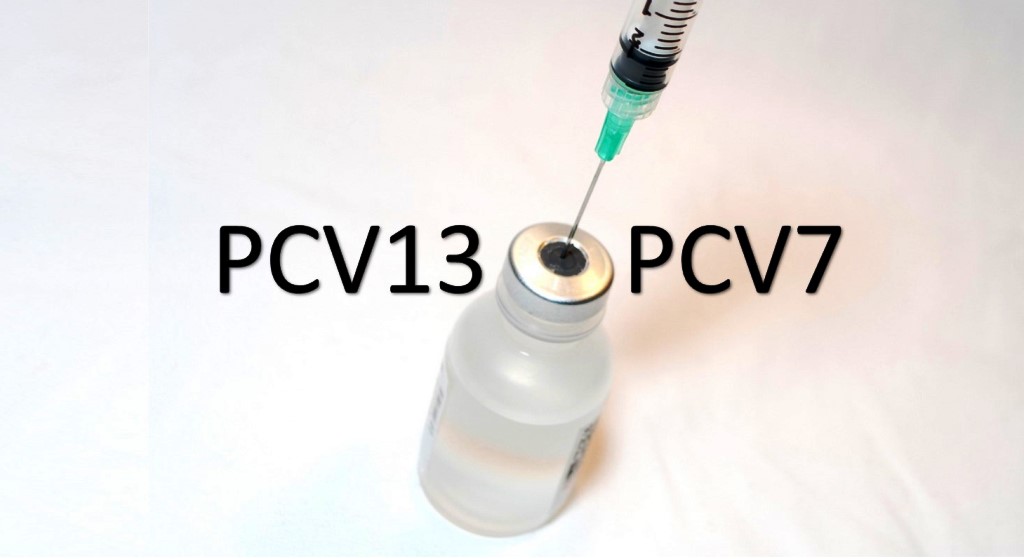
A comparison of 7-valent pneumococcal conjugate vaccine and its 13-valent counterpart suggests three primary doses with no booster yielded greater dividends for PCV7 than for PCV13, according to a recent study in Australia. “A randomized trial of PCV13 schedules found lower antibody responses to some serotypes after primary sequence of 2 doses compared with 3, highlighting the need for post-implementation studies in countries using different schedules to examine population level impact of PCV13,” Sanjay Jayasinghe,MBBS, MSc, from the National Centre for Immunization Research and Surveillance (NCIRS) in Australia, and colleagues wrote. “The Australian experience complements reports from other settings using alternate PCV schedules, in the context of continuing global considerations about the optimum use of pneumococcal conjugate vaccines in national programs.” Replacing universal PCV7 in Australia in 2011, PCV13 is administered at 2, 4 and 6 months (3+0 schedule). However, there are a lack of data on the impact of a timely...
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!