A joint research team from the A.N. Frumkin Institute of Physical Chemistry & Electrochemistry (RAS), NUST MISIS, and MIPT, as well as participants from several other research centers, have described the biophysical principles of influenza’s infiltration into the body’s cells.
The researchers have created a theoretical model describing the mechanical properties of the lipid membranes of both the virus and the targeted cells, making it possible to connect the mechanisms of membranes with cell resistance. A series of three articles containing the new research have been published in special editions of the International Journal of Molecular Sciences.
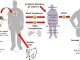
Viruses often present the main threat to the human body, with diseases like Human Immunodeficiency Virus (HIV), Herpes, Hepatitis, Ebola and forms of influenza, among others, causing serious damage. These viruses invade the cells of the affected body by merging their membrane with the cell membrane or its organelles. “Fusion proteins” facilitate this process, leading to contact between the lipid membrane of both the virus and the target cell.
As viruses are extremely simple constructions, only capable of parasitic functions and not capable of an independent production of energy, their fusion proteins can attack cell membranes only with their original energy. Essentially, the fusion proteins are like chains “shooting” into the membrane of the infected cell, trapping and pulling the cell membrane toward the virus to start the fusion process.
Because of this unusual mechanism of penetration, it turned out that the viral infection process of cells often depends on the properties of the cell’s lipid membrane, particularly its elasticity. In other words, the more difficult it is for viral proteins to mechanically deform the cell membrane, the less likely it is that the cell will be infected.
The researchers have developed a theoretical model that predicts the energy costs viral proteins need to overcome to cause the fusion of its own membrane with the “victim’s” membrane. Following the changes in the energy of this process, physicists have found several alternative attack scenarios – some of which end up with the virus “winning”, while other cases see a deadlock formed where the virus is no longer able to force itself upon a cell.
“We were inspired by experiments on viral fusion research using cryoelectronic microscopy methods. Unusual structures, not predicted by any of the existing theoretical models of membrane fusion, were found in them. That’s why we have proposed our modified fusion model and showed that the structures we detected are dead-end and lead to the virus merging with the cell. [We’ve also] calculated how the structure of viral fusion proteins affect the choice of the process’s pathway, whether successful or [not]”, said Timur Galimzyanov, a co-author of the research article and a researcher at the NUST MISIS Department of Theoretical Physics & Quantum Technologies.
It is noteworthy that all the conclusions described in the work were obtained using general approaches in Physics without the need to introduce specific chemical interactions, as often happens in cases such as these. Nevertheless, it turned out that the process of membrane fusion is influenced by several key factors, and a corresponding study was then devoted to each factor.
The geometric parameters of fusion proteins that attack the membrane of the “victim”-cell, the pH of the environment determining the structure of proteins, and the presence of so-called “rafts” are the three factors the researchers focused on. An article devoted to the third factor was published in mid-May 2018. That article explains how the “rafts”, islands of hard molecules, move relatively freely along the semi-liquid cell membrane and interact with viruses, a piece that plays a crucial role in determining how the fusion process will go. The result of this work was the relatively simple conclusion that the presence of the raft in the area of attack, at least in the case of HIV, promotes fusion, and, as a consequence, the successful penetration of viral genetic material into the cell.
The researchers emphasize that in the future their results will be adapted for a wider class of objects, although the theoretical model’s current predictions pose an important verification problem for the field. If the research’s conclusions about the role of a cell membrane’s mechanical properties are confirmed by future research, it could be the jumping off point in the study of viral processes and mechanisms of cell protection.
Source: The National University of Science and Technology MISiS