In the wake of the Ebola epidemic in West Africa, the World Health Organization (WHO) put forth a worldwide coalition to develop an R&D [Research and Development] Blueprint for Action to Prevent Epidemics. One focus of this blueprint involves creating R&D roadmaps for priority diseases.
The Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota in Minneapolis is working with WHO and the Wellcome Trust to create these roadmaps and providing information about the process via a series of webinars which will allow a wide range of stakeholders from the private and public sectors, as well as from government and academia, to participate in developing measures to help prevent and control priority diseases.
In a May 15th webinar hosted by CIDRAP, the Center’s director, Michael T. Osterholm, PhD, MPH, shared information about the process involved in developing an R&D roadmap for the priority pathogen Nipah virus.
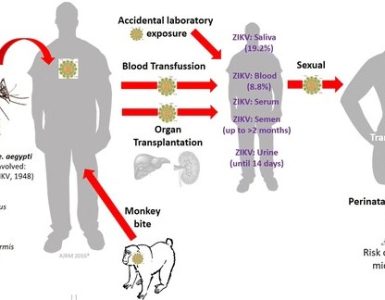
According to Dr. Osterholm, Nipah virus is an emerging infectious disease that is endemic in Southeast Asia. It is a zoonotic paramyxovirus, and predominantly spreads through contact with bats (in particular, fruit bats) or infected pigs. However, it can also be spread person-to-person. It causes severe disease in people, with high case fatality rates that have been reported to range from 40% to 100%.
Nipah virus was first identified in 1998-1999 in Malaysia and Singapore, in association with an outbreak of infectious respiratory and neurologic disease in farmed pigs. The pigs served as a source of infection among pig farmers and other people who were in close contact with the animals, resulting in encephalitis and respiratory illness in these individuals.
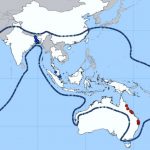
Although Nipah virus infection has not been identified in Malaysia or Singapore since 1999, outbreaks of Nipah encephalitis with high fatality rates have occurred on an almost annual basis in Bangladesh since 2001 (see Nipah Virus Distribution Map).
The roadmap for Nipah prioritizes the development of “robust medical countermeasures to detect, prevent, and control outbreaks” of infection in countries affected by the virus, said Dr. Osterholm. These measures include diagnostics, therapeutics, and vaccines.
According to the US Centers for Disease Control and Prevention, although ribavirin has been shown to be effective against Nipah virus in vitro, “human investigations to date have been inconclusive and the clinical usefulness of ribavirin remains uncertain.” Therefore, current treatment for the infection is limited to supportive care.
Dr. Osterholm stressed during the webinar that “roadmaps are pivotal in understanding markets for innovative products and identifying ways to leverage R&D investments, through coordinating research activities across a multitude of different partners.”
The timeline for the Nipah virus roadmap is currently on schedule. To date, a Nipah Roadmap Taskforce of 13 members have already participated in an expert review meeting and have continued to provide a review of the roadmap document.
The final draft of the roadmap document is anticipated in June, after incorporating additional feedback from stakeholders. However, the document will continue to be revised with input from WHO and other key partners before its final publication in August or September.
Source: Contagionlive.com