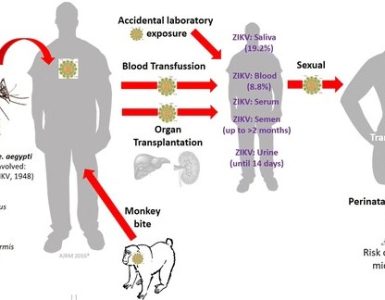
As Congress plays politics over emergency funds requested months ago, Zika is on the verge of spreading across the United States.
As Congress plays politics over emergency funds requested months ago, Zika is on the verge of spreading across the United States.
When an epidemic is imminent, vaccines can’t be developed quickly enough because assuring their efficacy and safety requires trials that take months to years to complete.Candidate vaccines for Zika won’t begin large-scale trials until 2017 and, assuming they work safely, would not be available until 2018 or later.
The next best thing is widespread testing of at-risk populations so new cases can be identified immediately and prevented from transmitting to others. Widespread testing can also pinpoint “hot spots” where community-wide measures like mosquito spraying should be targeted. This strategy mirrors what Tony Fauci of the National Institutes of Health (NIH) has outlined for stamping out HIV/AIDS, for which there is also no vaccine.
Authorities in Miami are attempting this approach with door-to-door testing of Zika in affected neighborhoods. Widespread testing at greater scale however — as may soon be needed throughout Florida and elsewhere — requires a point-of-care doorstep diagnostic that doesn’t depend on specialized equipment and personnel and can be deployed quickly and widely to test people at their homes.
Currently, however, most pandemic-prone diseases, including Zika, are diagnosed by polymerase chain reaction (PCR), a technique that requires special laboratories and highly trained technicians. These are difficult to scale or decentralize. The United States does not have enough PCR capacity to screen every pregnant woman — approximately 2% of the total population.
Though the Florida government is officially committed to free testing for all pregnant women, capacity is limited and some women must pay $150 to $500 to get tested at private labs. This gap exposes systemic vulnerabilities to pandemic threats and bioterrorism. If we were facing a pathogen more widespread than Zika, how could we test 100% of the population if we cannot even screen 2%?
Point-of-care diagnostics, if prioritized, could be developed within months at a cost of tens of millions of dollars as compared to the hundreds of millions of dollars required for vaccines. For Zika, the need for doorstep diagnostics has been acknowledged, but progress has been slow. There is no federal agency with the responsibility and authority to coordinate industry, academia, and other agencies and drive forward the necessary development. Instead, innovation is left to individual researchers and companies to pursue on their own with the government waiting passively like a food critic for solutions to be offered for review. For example, our colleague who developed the first Ebola doorstep diagnostic authorized by the U.S. Food and Drug Administration (FDA) has made progress on a Zika test but needs more Zika blood samples to refine the technology. His numerous inquiries to multiple government agencies — each of whom redirects him to another — have led only to dead ends all around.
The FDA has an accelerated pathway for “emergently needed” diagnostics, but relies on developers to submit results from studies they arrange themselves. Without a standard methodology or designated organization for conducting these evaluations, even after data is submitted to the FDA and authorization is granted, other agencies, such as the U.S. Centers for Disease Control and Prevention (CDC) or the World Health Organization (WHO), may question the accuracy and conditions under which those results were achieved, raising doubts about new tests and stalling their deployment even during an emergency. This is exactly what happened during Ebola. A doorstep diagnostic that showed promising results as early as October 2014 was not authorized by the FDA until January 2015 — and it was never used in the field. In retrospect, that test could have averted thousands of infections and ended the epidemic sooner.
All of this begs the question: Who is responsible for the development of a doorstep diagnostic for Zika? If not WHO, CDC, the U.S. Biomedical Advanced Research and Development Authority, NIH, or FDA, whose job is it to ensure that sufficient support is available and roadblocks are removed so new diagnostics get developed quickly during a rapidly expanding pandemic?
We need a single government agency or, as former Ebola Czar Ron Klain suggested, a pandemic directorate of the National Security Council that is empowered and held accountable for proactively fast-tracking the development of new diagnostics from academia, industry, and within government. We also need “grand challenge” prize grants (e.g., $100 million) to incentivize these efforts. Without sufficient incentives, many developers and companies deem the investment needed to develop diagnostics that may not find a market once the pandemic is over too risky financially.
With a vaccine nowhere on the horizon, we ignore Zika at our own peril. Institutional reforms are needed to ensure a stronger pipeline for diagnostics essential to stopping this and future pandemics.
Source: Harvard Business Review