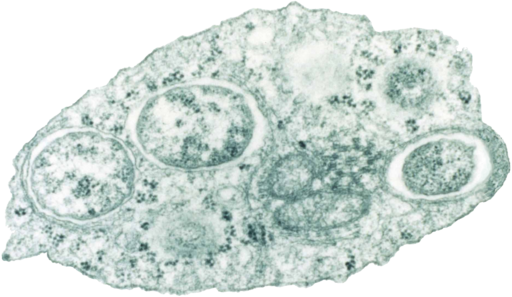
A strategy for fighting dengue fever with bacteria-armed mosquitoes has passed its most rigorous test yet: a large, randomized, controlled trial. Researchers reported dramatic reductions in rates of dengue infection and hospitalization in areas of an Indonesian city where the disease-fighting mosquitoes were released. The team expects the World Health Organization (WHO) to formally recommend the approach for broader use. The findings are a “breakthrough” that brings the approach “much closer to … being an official strategy to control dengue,” says Ewa Chrostek, an infection biologist at the University of Liverpool who was not involved with the work. WHO estimates there are 100 million to 400 million infections per year with dengue, which can cause high fever and severe joint pain. The bacterium Wolbachia pipientis naturally inhabits many insects, though not Aedes aegypti mosquitoes, the main transmitter of dengue virus. In A. aegypti cells, the bacterium can block viruses, including dengue, from...
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!