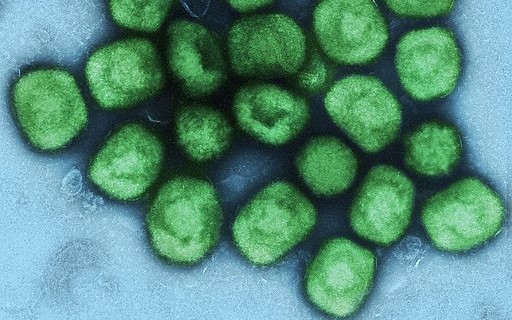
Favored shot is a seemingly safer smallpox vaccine, but efficacy remains unclear In 1959, German microbiologist Anton Mayr took a strain of vaccinia, a poxvirus used to inoculate against smallpox, and started to grow it in cells taken from chicken embryos. After several years of transferring the strain to fresh cells every few days, the virus had changed so much it could no longer reproduce in most cells from mammals. But it could still produce an immune response that protected against smallpox. Mayr had set out to study how poxviruses evolve, but by accident he had produced a potentially safer smallpox vaccine. Dubbed Modified Vaccinia Ankara (MVA) because the original viral strain came from that Turkish city, the vaccine had a short career. “With smallpox eradicated in 1980, it disappeared into the freezer,” says Gerd Sutter, a virologist at the Ludwig Maximilian University of Munich, who has studied Mayr’s vaccinia strain for...
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!