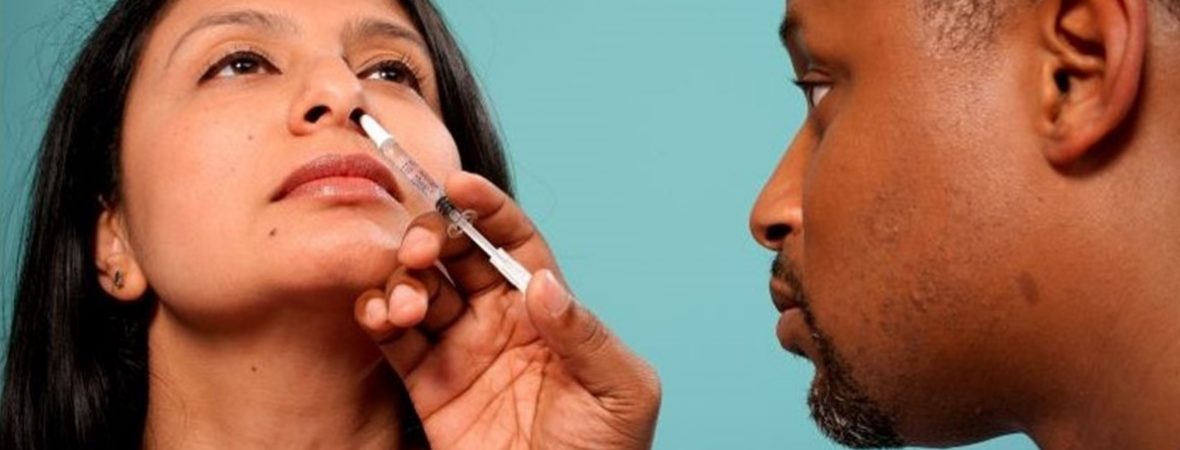
Public health experts believe that intranasal vaccines for coronavirus, administered through nose rather than muscles, can be a potential gamechanger in India’s fight against the coronavirus pandemic. The nasal vaccine is different from two intramuscular vaccines which recently got approval, as it is non-invasive, needle-free, doesn’t require trained healthcare workers, eliminates needle-associated risks, suits children and adults and has scalable manufacturing. Several studies have proved that the clinical efficacy of intranasal vaccine is superior to that of injectable vaccine. In India, Hyderabad-based Bharat Biotech has sought Drugs Controller General of India’s (DCGI) approval to conduct clinical trials of its nasal vaccine for coronavirus. Last month, Pune-based Serum Insititute of India and Codagenix Inc had announced that they have received regulatory approval in the United Kingdom to begin an early-stage trial of their single-dose, intranasal coronavirus vaccine. “At the moment, four vaccines are undergoing phase 1 and 2 of nasal vaccine trials in the world....
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!