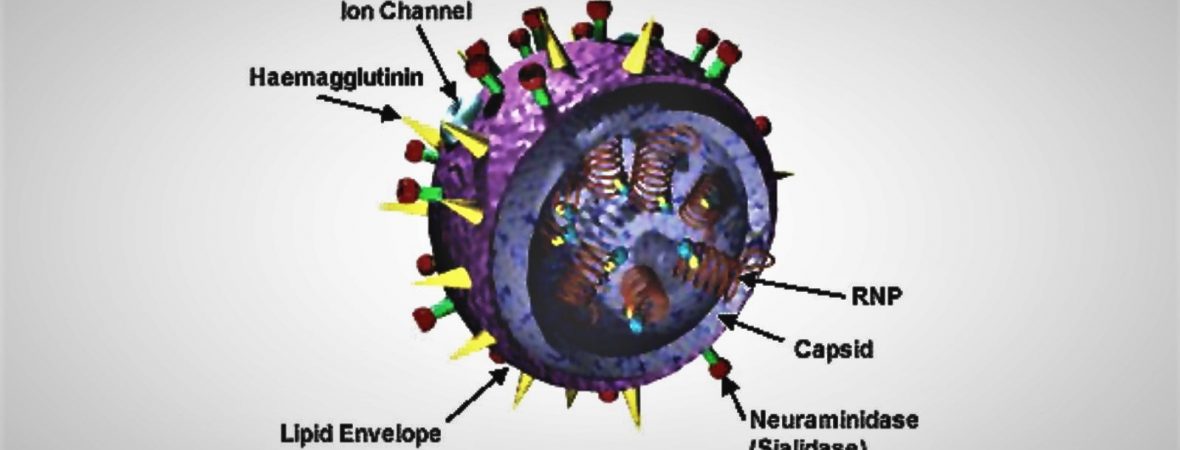
The influenza A viruses, which are responsible for deadly pandemics in the past, still remain a major global public health problem today. Molecules known as virulence factors are produced by bacteria, viruses, and fungi to help them to infect host cells. One of virulence factors found in the influenza A viruses is hemagglutinin (HA). Researchers at Kanazawa University have recently studied the structure of HA of avian influenza virus, H5N1, using high-speed atomic force microscopy (HS-AFM). Their findings are essential for developing therapeutic approaches against influenza A viruses in future. HA is initially synthesized by host cells in its precursor form known as HA0. Conversion of HA0 to HA depends on the pathogenicity of influenza A viruses: extracellular conversion for low pathogenic influenza A viruses and intracellular conversion for highly pathogenic influenza A viruses. Therefore, understanding the structure and properties of HA0 is paramount to deciphering HA. Richard Wong and his...
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!