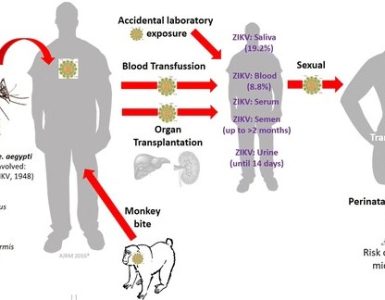
An experimental vaccine against the Zika virus reduced the amount of virus in pregnant rhesus macaques and improved fetal outcomes, according to a study published in the journal Science Translational Medicine.
The study marks the first test of a Zika vaccine given before conception with exposure to the virus during pregnancy, mimicking a real-world scenario where women could be vaccinated months or years before becoming pregnant.
The scientists from U.S. National Institute of Allergy and Infectious Diseases vaccinated 13 female monkeys with VRC5283, the experimental Zika DNA vaccine, before their pregnancy. Then they exposed them to Zika virus at intervals representing first and second trimesters.
The vaccinated females had less virus in their blood and the virus persisted for a shorter duration after exposure. Also, two unvaccinated animals lost the fetus early in pregnancy while there was no early fetus loss in the vaccinated group.
At the end of the pregnancy, 11 of 12 fetuses in the unvaccinated control group had detectable Zika virus RNA while no Zika virus RNA was detected in the 13 fetuses from the vaccinated group.
It suggests that antibodies against Zika virus in the vaccinated mother animals correlated with protection against the virus.
The candidate vaccine is now in global phase II trials to test its safety and ability to elicit an immune response in humans. Results from this animal studies can help support the case for approving the vaccine, while additional clinical trials are needed to determine its efficacy.
Source: xinhuanet.com