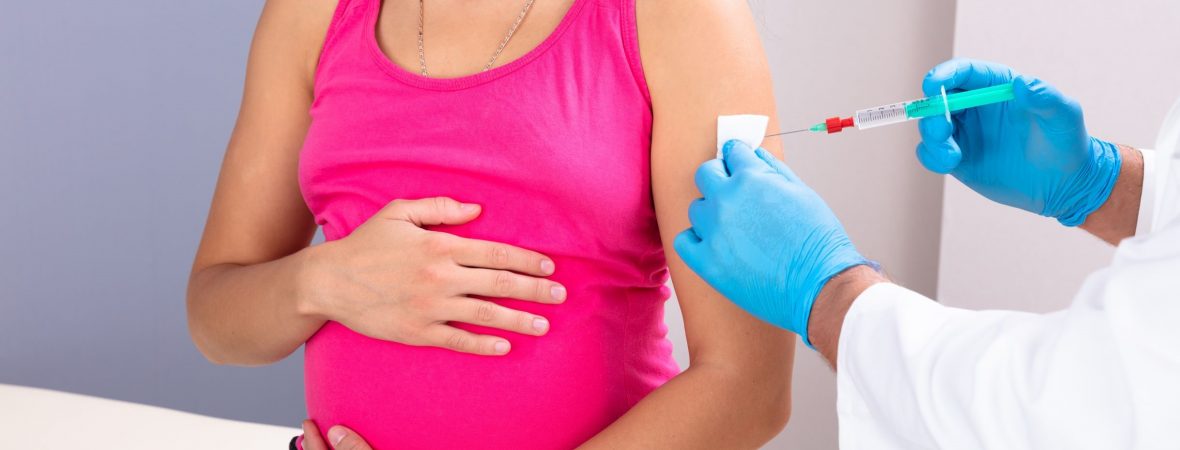
Influenza vaccination can be underused among pregnant women, despite evidence it can protect both the mother and infant from the flu. A team, led by Fatimah S. Dawood, MD, Influenza Division, Centers for Disease Control and Prevention, quantified the risk of antenatal influenza and examined its association with perinatal outcomes. In the prospective cohort, the investigators examined 11,277 pregnant women in India, Peru, and Thailand prior to the 2017 and 2018 influenzas seasons, of which 1474 (13%) received influenza vaccines. Each participant was at least 18 years old with expected delivery dates 8 weeks or more following the start of the influenza season. The median age of the patient population was 26 years old and the gestational age was 19 weeks. A total of 310 patients had influenza (3%), 270 of which had influenza A (87%) and 40 of which had influenza B (13%). The researchers contacted the women twice a week until the...
🔒 Premium Content - For Free
Unlock this content by becoming a Global Health Press subscriber. Join for exclusive articles, expert research, and valuable insights!